|
The alkali metal sodium (atomic number 11) has one more electron than the neon atom. Neon, with its configuration ending in \(2s^2 2p^6\), has eight valence electrons. In fact, the number of valence electrons goes up by one for each step across a period until the last element is reached. The diagram below shows the number of valence electrons (VE) for the main group elements. How many valence electrons does boron have? You must recognize that the second principal energy level consists of both the \(2s\) and the \(2p\) sublevels and so the answer is three. This pattern allows us to quickly identify the number of valence electrons for main group elements on the periodic table, even if we don't have Bohr models to reference. Elements that have similar chemical properties are grouped in columns called groups (or families). The distance between the nucleus and the electron in 3p is further than the 3s2 electrons. Common chemical compounds are also provided for many elements. Comprehensive data on the chemical element Magnesium is provided on this page including scores of properties, element names in many languages, most known nuclides of Magnesium. Therefore the 3p1 electron is subject to electron-electron repulsion by 3s2 electrons. Periodic Table of Elements Element Magnesium - Mg. The number of valence electrons depends on the octet rule. Elements within the same group share the same number of valence electrons. Atoms in a period have the same number of electron shells. Lithium has a single electron in the second principal energy level and so we say that lithium has one valence electron. When you add 3p1, the outermost electron is in the 3p orbital (even though 3s and 3p are all valence electrons). The number of valence electrons of an element can be determined by the periodic table group (vertical column) in which the element is categorized. An element period is a horizontal row on the periodic table. In the second period elements, the two electrons in the \(1s\) sublevel are called inner-shell electrons and are not involved directly in the element's reactivity or in the formation of compounds. Valence electrons are the electrons in the highest occupied principal energy level of an atom. This octet rule holds for elements in the second and third periods (or rows) of the periodic table. In the study of chemical reactivity, we will find that the electrons in the outermost principal energy level are very important and so they are given a special name. This need to gain a filled valance electron shell by having 8 valence electrons is known as the octet rule and explains why certain elements are stable or unstable despite being electrically neutral. The order of filling subshells is the same: 1s, 2 s, 2 p, 3 s, 3 p, 4 s, 3 d, 4 p, 5s, 4d, 5p, 6s, etc. Oxygen will gain 2 electrons.\( \newcommand\) provides a simple way to remember the order of filling the subshells in determining the electron configuration. The second shell has six electrons ( 2 s 22 p 4) and needs two electrons to achieve octet. The electron configuration of O atom is 1 s 22 s 22 p 4. How many electrons must O lose/gain to achieve octet? Write the formula of the resulting ion and its electron configuration. Write the electron configuration of oxygen atom (Z=8). 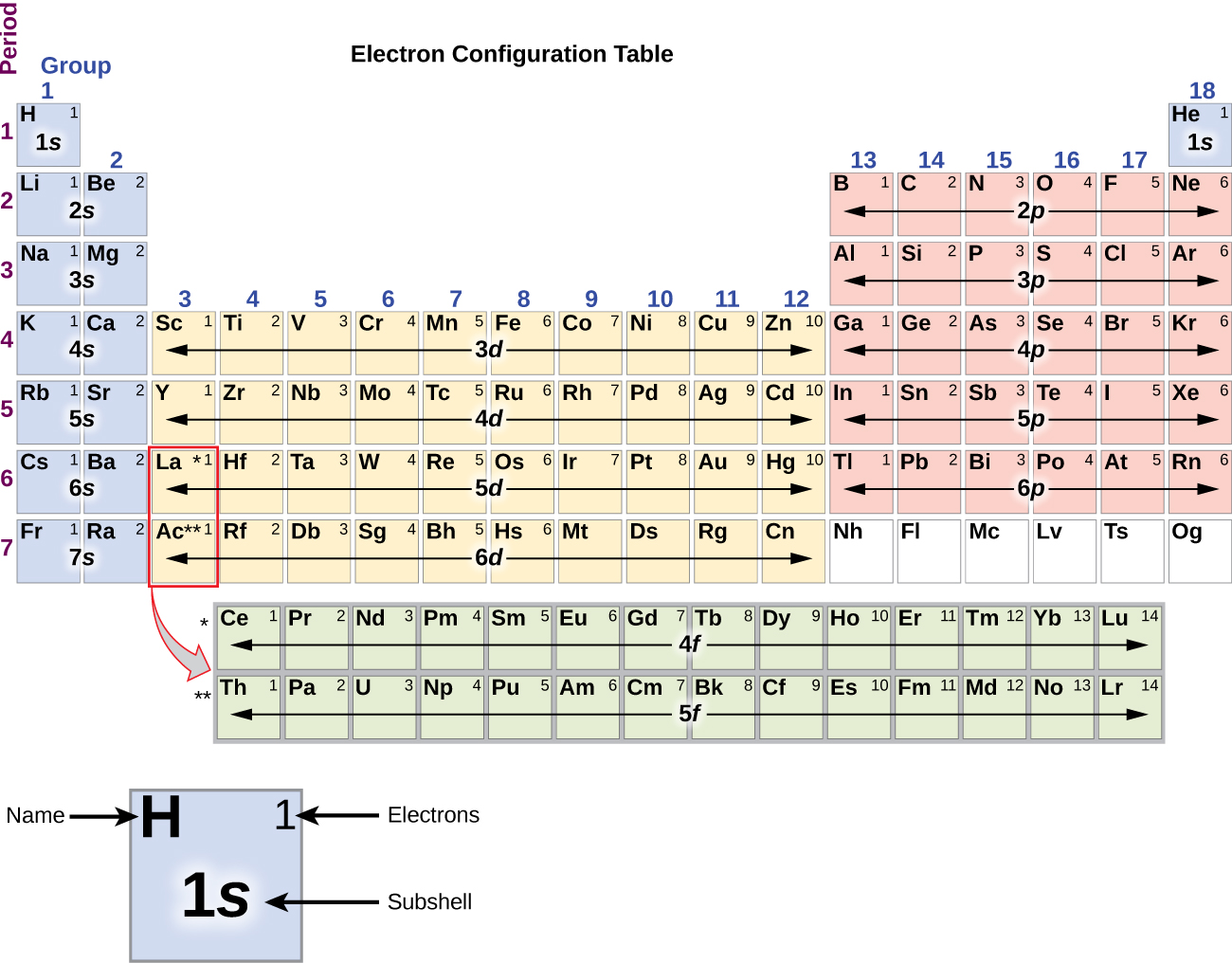
In macroscopic samples of sodium chloride, there are billions and billions of sodium and chloride ions, although there is always the same number of cations and anions. 
The number of electrons lost by the sodium atom (one) equals the number of electrons gained by the chlorine atom (one), so the compound is electrically neutral. Notice that there are no leftover electrons. The resulting combination is the compound sodium chloride. With two oppositely charged ions, there is an electrostatic attraction between them because opposite charges attract. On the right, the chloride ion has 18 electrons and has a 1− charge. On the left, the chlorine atom has 17 electrons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
